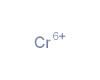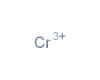The largest block of elements in the periodic table is a group known as the transition metals. These metals are found in groups three through twelve of the periodic table (the so-called d-block elements), although there are ongoing differences of opinion about exactly which elements should be classed as transition metals and which should not. Some of the most commonly used metals on Earth are transition metals, including zinc, titanium, copper and iron. Precious metals like silver and gold are also transition metals. The transition metals typically have the following properties: a tendency to form coloured compounds, good conductors of heat and electricity, high melting and boiling points, high density, malleable (can be bent or hammered into shape), ductile (can be drawn into wires), relatively hard, less reactive than alkali and alkaline earth metals. Most transition metals have several possible oxidation states. The maximum oxidation state possible will in many cases be equal to the number of unpaired electrons in the (n-1)d orbitals, plus two (the number of electrons usually found in the ns orbital) (Wells, 2017). In our database the following metals are classified as transition metals: Scandium (Sc)*, Titanium (Ti) Vanadium (V)*, Chromium (Cr), Manganese (Mn), Iron (Fe), Cobalt (Co*), Nickel (Ni) , Copper (Cu), Zinc (Zn), Yttrium (Y)*, Zirconium (Zr), Niobium (Nb)*, Molybdenum (Mo), Technetium (Tc), Ruthenium (Ru)*, Rhodium (Rh)*, Palladium (Pa)*, Silver (Ag), Cadmium (Cd), Hafnium (Hf)*, Tantalum (Ta)*, Tungsten (W)*, Rhenium (Re), Osmium (Os)*, Iridium (Ir)*, Platinum (Pt)*, Gold (Au), Mercury (Hg), Rutherfordium (Rf), Seaborgium (Sg), Bohrium (Bh), Hassium (Hs), Meitnerium (Mt), Ununnilium, Unununium, Ununbium
“Post-transition metals are a set of metallic elements in the periodic table located between the transition metals to their left, and the metalloids to their right. Depending on where these adjacent groups are judged to begin and end, there are at least five competing proposals for which elements to include: the three most common contain six, ten and thirteen elements, respectively. All proposals include gallium, indium, tin, thallium, lead, and bismuth. Physically, post-transition metals are soft (or brittle), have poor mechanical strength,and have melting points lower than those of the transition metals. Being close to the metal-non-metal border, their crystalline structures tend to show covalent or directional bonding effects, having generally greater complexity or fewer nearest neighbours than other metallic elements. Chemically, they are characterised—to varying degrees—by covalent bonding tendencies, acid-base amphoterism and the formation of anionic species such as aluminates, stannates, and bismuthates (in the case of aluminium, tin, and bismuth, respectively). They can also form Zintl phases (half-metallic compounds formed between highly electropositive metals and moderately electronegative metals or metalloids). Alternate names for this group are B-subgroup metals, other metals, and p-block metals; and at least eleven other labels.“ (Wikipedia) In our database the following metals are classified as post-transition metals: aluminum (Al), gallium (Ga)*, indium (In)*, thallium (Tl), Tin (Sn), lead (Pb) and bismuth (Bi)*.
* on the 2017 CRMs (27) list
References
Wikipedia, Available from: https://en.wikipedia.org/wiki/Post-transition_metal, Accessed: 21st of June 2018
Wells, Christopher J. (2017) TechnologyUKnet, Alkali and Alkaline Earth Metals, Available from: https://www.technologyuk.net/physics/matter/transition-metals.shtml, Accessed: 21st of June 2018
CRMs (27) (2017) COMMUNICATION FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT, THE COUNCIL, THE EUROPEAN ECONOMIC AND SOCIAL COMMITTEE AND THE COMMITTEE OF THE REGIONS on the 2017 list of Critical Raw Materials for the EU
Photo: http://chemed.chem.purdue.edu/genchem/topicreview/bp/ch12/trans.php